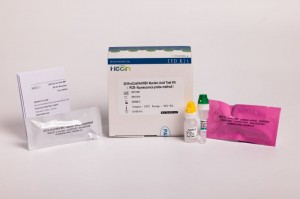
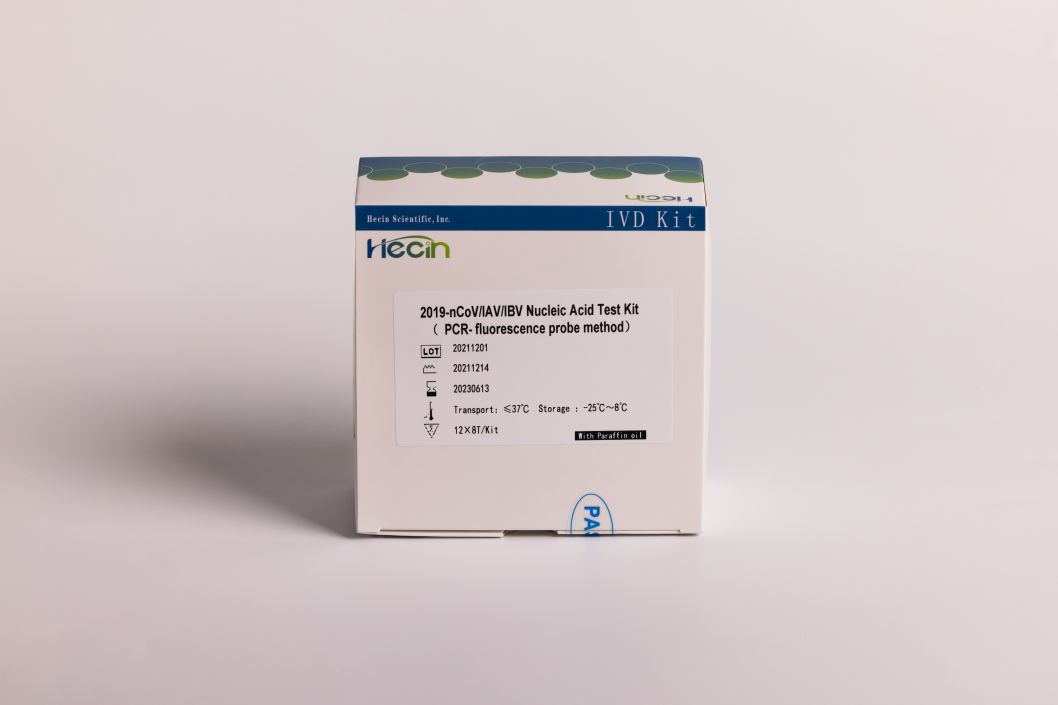
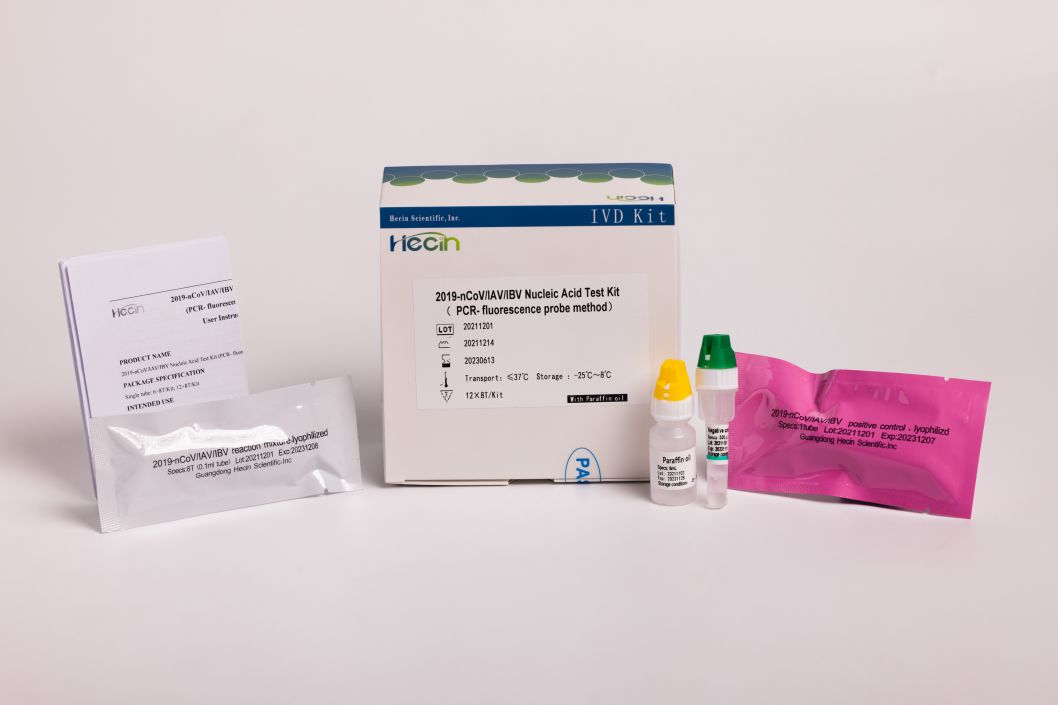
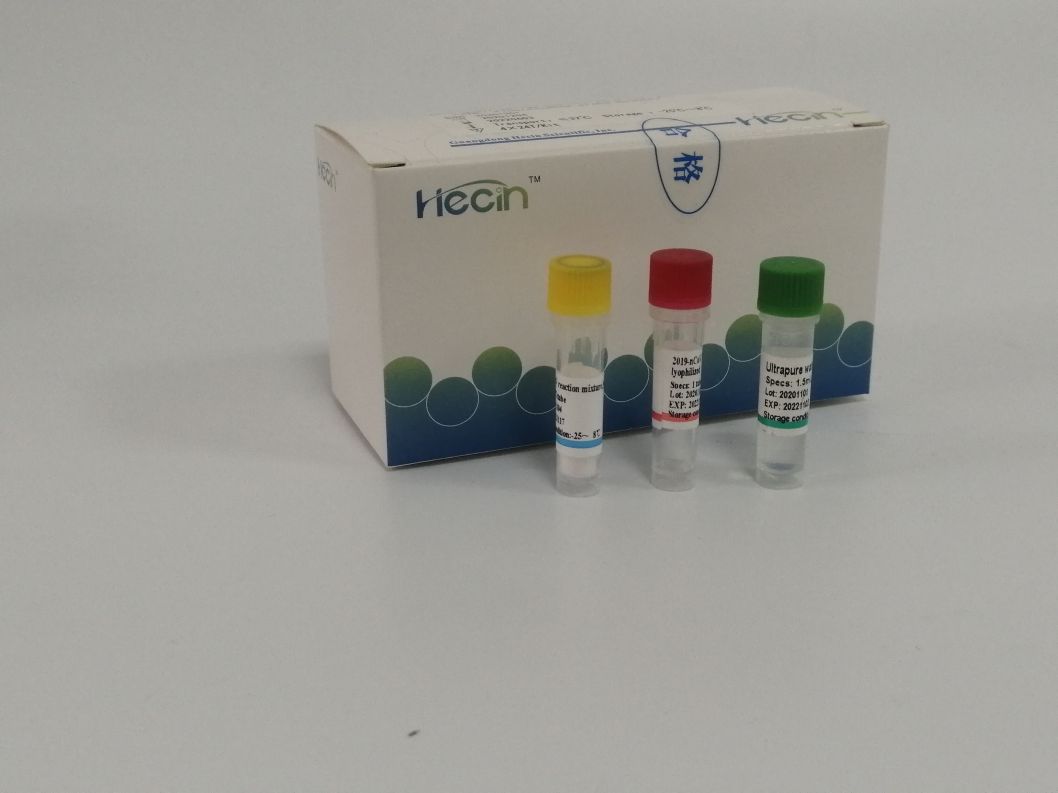
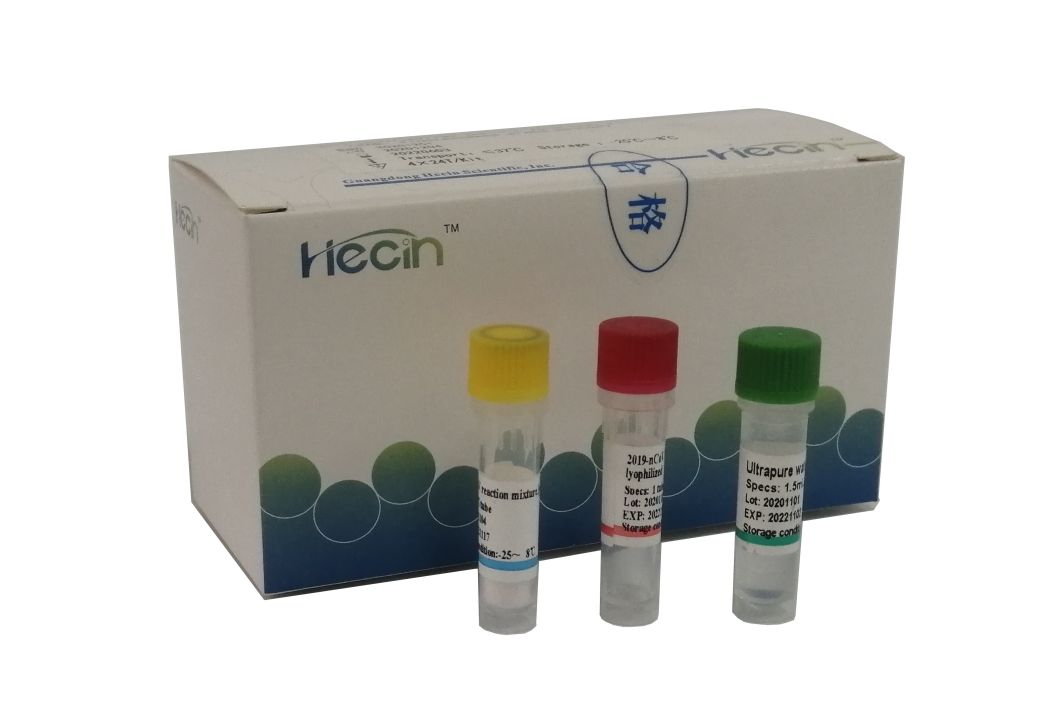
2019-nCoV/IAV/IBV Nucleic Acid Test Kit (PCR- fluorescence probe nzira)
Chinangwa Kushandiswa
Iyi kit ndeye chaiyo-nguva RT-PCR bvunzo yakanangana nekuonekwa kwemhando yeRNA kubva ku2019-nCoV, Influenza A virus (IAV), Influenza B virus (IBV).
Zvakanakira
Kuonekwa kwekubatana:Kuonekwa kwemhando yeRNA kubva ku2019-nCoV, Influenza A virus (IAV), Influenza B virus (IBV).
Kufambisa mukati me37 ℃ kwemwedzi mitatu:lyophilized reagent yakanyanya kugadzikana; Mamiriro ekufambisa: ≤37 ℃, yakagadzikana kwemwedzi mitatu.
Kushanda kushoma:Iyo amplification reaction solution yakasanganiswa pamwe chete, kuderedza kushanda kwerabhoritari.
Mhando dzakasiyana dzemuenzaniso:Specimen type nasopharyngeal swab; oropharyngeal swabs;sputum;alveolar lavage fluid specimens.
Kuita kwakavimbika:Coincidence mwero wezvakanaka kana zvisina kunaka zvinongedzo zvigadzirwa: 100%.iyo coefficient yekusiyana kweCt value (CV, %) ishoma pane kana kuenzana ne5%.
Zviridzwa
UltraFast QPCR FQ-8A,QuantGene 9600 Real-Time PCR System ;ABI 7500 Real-Time PCR System, Roche LightCycler96 Real-Time PCR System, Tianlong TL988 Real-Time PCR System, SLAN Real-Time PCR System nezvimwewo.
Fluorescent channel inodiwa
N gene yekuona chiteshi: FAM
IBV yekuona chiteshi: VIC
IAV yekuona chiteshi: Tex Red
Internal control yekuona chiteshi: CY5
Muganhu Wekuonekwa
2019-nCoV : 400 makopi/mL.
Influenza A virus: 1.5 TCID50/mL.
Hutachiona hwefuruwenza B: 2.0 TCID50/mL.
Analytical chaiyo
Iyi test kit inogona kushandiswa kuona 2019-nCoV, Influenza A virus (2009 H1N1, H1N1, H3N2, H5N1, H7N9), Influenza B virus (Yamagata, Victoria) kunyanya.



Product specification
12X8T
12X4T
HS28- 12X8T/Kit
HS28- 4X24T/Kit